

 Secure and encrypted payment processing
Secure and encrypted payment processing We ship to over 40 countries including the USA, UK, Europe, Australia and Japan
We ship to over 40 countries including the USA, UK, Europe, Australia and Japan Guaranteed refund or reship if you haven't received your order
Guaranteed refund or reship if you haven't received your order
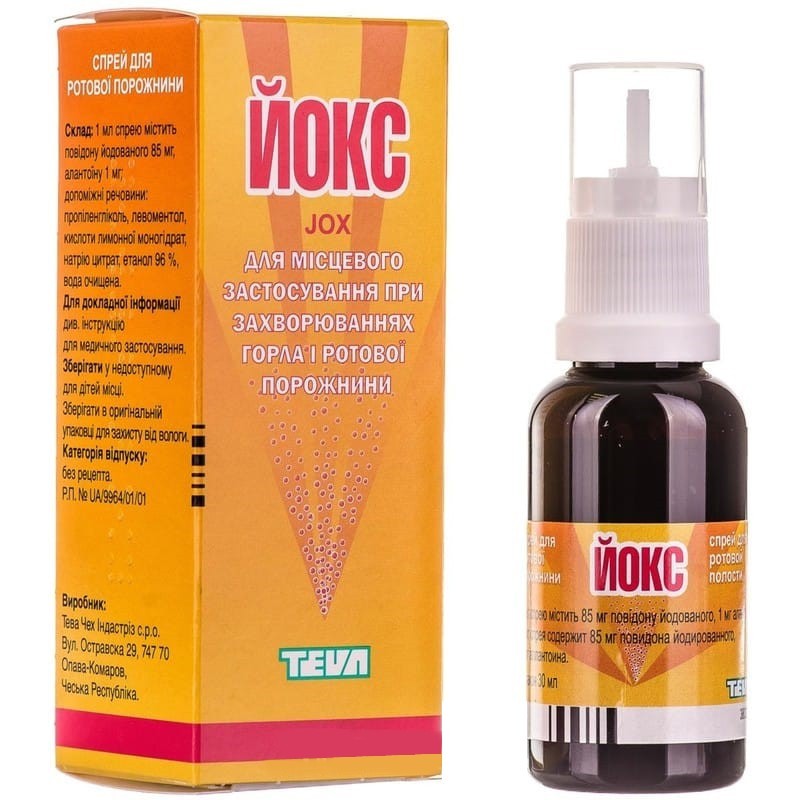
YOX (JOX)
COMPOSITION AND FORM OF ISSUE:
rinse. d / throat, conc. fl. 50 ml, No. 1
| Povidone iodine | 85 mg / ml |
| Allantoin | 1 mg / ml |
No. UA / 1829/02/01 from 05/06/2010 to 05/06/2015
spray fl. with spray 30 ml
| Povidone iodine | 85 mg / ml |
| Allantoin | 1 mg / ml |
Other ingredients: propylene glycol, levomenthol, citric acid monohydrate, sodium dihydrocytrate, ethanol, purified water.
No. UA / 9964/01/01 from 08.25.2009 to 08.25.2014
PHARMACOLOGICAL PROPERTIES:
The main active substance of the drug Yoks is povidone-iodine. The specified substance releases iodine when it comes into contact with the skin or mucous membrane, which has a wide spectrum of antimicrobial action due to the direct effect on the proteins of microorganisms. Yoks has a partial bactericidal effect on gram-positive and gram-negative cocci and bacilli, on some anaerobic bacteria, yeast or saccharomycetes, simple viruses. Yox also contains allantoin, which has an anti-inflammatory effect. Due to the combined action of allantoin with povidone iodine, the therapeutic effect of the drug is manifested.
With topical application of povidone iodine to intact skin, iodine absorption is minimal. When used in the oral cavity, a more pronounced systemic absorption of iodine is noted, as a result of which interference with thyroid function can occur. Iodine in the body turns into iodides, which accumulate mainly in the thyroid gland and are secreted mainly by the kidneys, as well as in small amounts with feces, saliva, and sweat. Iodides pass through the placental barrier, penetrate into breast milk.
INDICATIONS:
angina catarrhal, follicular, lacunar; strep throat caused by streptococcus (with simultaneous treatment with antibiotics); chronic tonsillitis; pharyngitis, glossitis, aphthous stomatitis; initial flu symptoms.
APPLICATION:
spray: applied topically to the mucous membrane of the oral cavity and pharynx. Remove the protective cap and fit the applicator. Press the applicator 2–3 times so that the solution enters the sprayer and splashes after pressing. After that, insert the applicator tube 2-3 cm into the oral cavity, close the mouth, hold your breath and press 1-3 times to the right side, and then to the left side. Apply to adults and children over 8 years old, as a rule, 2–4 times a day.
Throat conditioner, concentrate. Diluted solution is applied topically to the mucous membrane of the oral cavity and pharynx for rinsing or rinsing. Do not swallow the drug.
Yoks concentrate is diluted with drinking water in a ratio of 1: 20–1: 40 (namely: 2.5–5 ml when using a measuring cap or ½ teaspoon per 100 ml of water). Rinse or rinse the oral cavity and pharynx with 10-15 ml of diluted solution for about 30 s. Apply to adults and children over 6 years of age 2–4 times a day, no more than 6 times a day, with an interval between two applications of at least 4 hours.
Elderly patients are shown the same doses as adults.
There are no data on specific geriatric cases with local use of iodine.
The duration of treatment depends on the nature and course of the disease, which is determined by the doctor.
CONTRAINDICATIONS:
hypersensitivity to iodine and other components of the drug, hyperthyroidism, decompensated heart failure, renal failure, pregnancy and lactation, preparation for research or treatment with radioactive iodine (2 weeks before and after); in the shape of mouthwash, concentrate - children under the age of 6 years. In the shape of spray - children under the age of 8 years.
SIDE EFFECTS:
the drug is well tolerated. Allergic reactions to iodine may occur (itching, hyperemia, urticaria, dry mouth). These reactions are usually transient. Symptoms of hypersensitivity were very rare.
Side effects are listed below in frequency of occurrence: very often: ≥ 1/10. Often: ≥ 1/100 to <1/10. Infrequently: ≥ 1/1000 to <1/100. Rarely: ≥ 1/10 000 to <1/1000.Very rare: <1/10 000.
From the immune system: very rarely - anaphylactic shock.
From the gastrointestinal tract: rarely - dry mouth.
On the part of the skin and subcutaneous tissue: rarely - urticaria. Very rarely - angioedema.
General disorders and reactions at the injection site: rarely - burning sensation at the site of use (especially in children).
Injuries, poisoning and complications during the procedures: iodism (metallic taste, increased salivation, swelling of the eyes or larynx, pulmonary edema, skin rash, gastrointestinal disorders) after prolonged use.
Systemic side effects: metabolic acidosis, hypernatremia, impaired renal function, diarrhea (after excessive absorption of iodine).
SPECIAL INSTRUCTIONS:
it is impractical to simultaneously use the drug Yoks with other drugs intended for use in the oral cavity and throat, especially with hydrogen peroxide.
With laryngitis, the drug can be used only in exceptional cases.
Use with caution in patients with renal failure. In such patients, the level of transaminases may increase, therefore, monitoring of transaminase parameters in blood plasma is necessary.
It is necessary to avoid contact of the drug with the mucous membrane of the eyes. If the solution gets into the eye, rinse it with water.
If symptoms of hypersensitivity or local irritation with the use of the drug do not disappear, you should stop taking it.
The drug can stain the mucous membrane of the oral cavity, teeth, skin and clothing. Stains on the mucous membrane of the mouth, teeth and skin can be removed with ethanol. If within 3 days the patient’s condition does not improve or adverse reactions, unusual sensations are noted, it is necessary to consult a doctor.
The drug contains ethanol.
During pregnancy and breastfeeding
The active substance of the drug crosses the placental barrier and is excreted in breast milk, so the drug is not recommended for use during pregnancy and lactation.
Children
The drug is used in children over the age of 6 years.
Spray: It is used to treat children over 8 years old. For children aged 8 to 12 years, the drug is used under the supervision of a doctor. Children should use the drug with caution, given the possibility of laryngospasm.
Influence on the ability to drive vehicles or other mechanisms. Does not affect.
INTERACTIONS:
It is not recommended to combine the drug with other antiseptic agents for the oral cavity and pharynx, primarily with hydrogen peroxide.
Concomitant use with lithium preparations has an additional hypothyroid effect.
It is not recommended to use povidone iodine with preparations containing the following substances: chlorheximide, silver sulfadiazine, alkaloids and mercury.
The use of the drug leads to false positive results of a blood test and a test for determining blood culture. When studying thyroid function, protein-binding iodine may increase.
Iodine is incompatible with ammonia; salts of iron, bismuth, copper, lead and mercury; potassium chlorate and other oxidizing agents; mineral acids; strychnine hydrochloride; quinine sulfate and other alkaloid salts.
OVERDOSE:
in case of ingestion of the drug, acute poisoning developed - a metallic taste was felt in the mouth, digestive disorders - vomiting, stomach pain, diarrhea. Anuria, glottis edema followed by asphyxiation, aspiration pneumonia, or pulmonary edema were noted within 1-3 days. In some cases, significant circulatory disorders were noted. Symptomatic treatment: standard measures to prevent absorption of the drug - ingestion of activated carbon and 1-5% solution of Na-thiosulfate to convert iodine to iodide. Milk and starch mucus are indicated.The elimination of absorbed iodine can be accelerated by osmotic diuretics. There is no specific antidote.
STORAGE CONDITIONS:
in the dark place at a temperature of no higher than 25 ° C.